Order of atomic orbitals
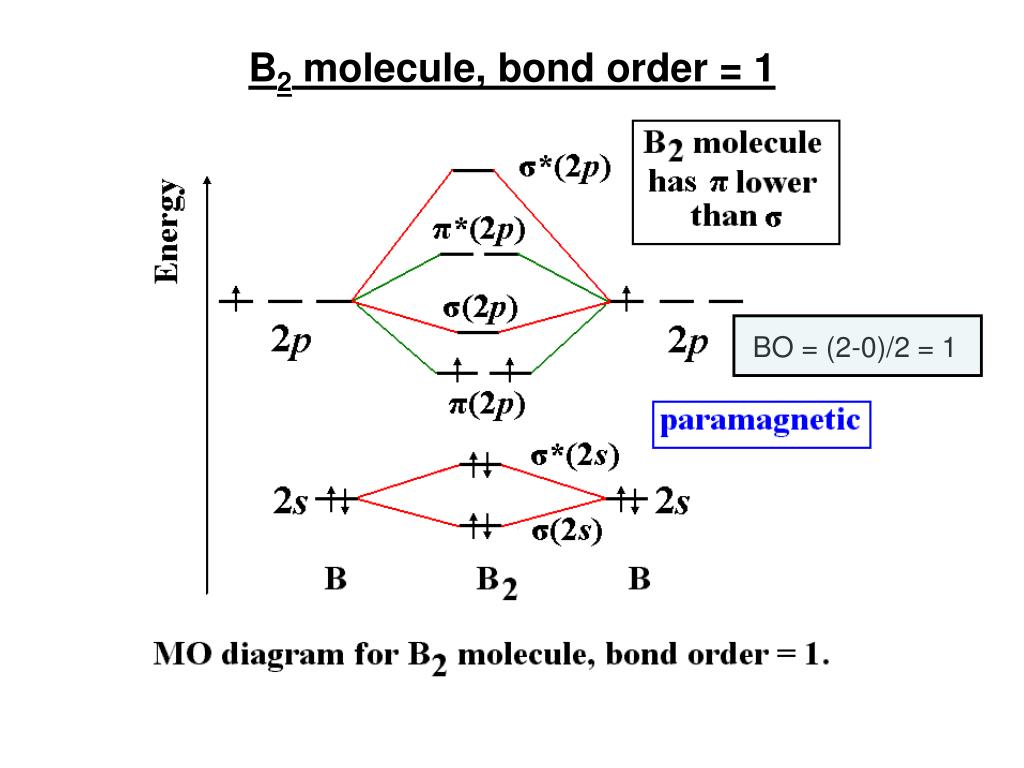
Within each shell of an atom there are some combinations of orbitals. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental). For lower elements, the difference between the energy levels of different orbitals is larger and exceptions are not commonly observed. 2) Orbitals are combined when bonds form between atoms in a molecule. Subsequently, when l6, the name of the atomic orbital will be 'I' and when l7, the name of the atomic orbital will be 'k'. Each electron is represented as either an up-facing arrow or a down-facing arrow. In the fields of quantum mechanics and atomic theory, these mathematical functions are often employed in order to determine the probability of finding an electron (belonging to an atom.
#ORDER OF ATOMIC ORBITALS FULL#
This is because of extra stability of half- filled and full filled orbitals. It very well may be noticed that the next atomic orbitals can be named one after another in order, precluding the letter 'j' (which is done in light of the fact that specific dialects don't recognize the letters 'j' and 'I'). The electrons are drawn in on blanks representing the orbitals. Atomic orbitals are mathematical functions that provide insight into the wave nature of electrons (or pairs of electrons) that exist around the nuclei of atoms. However, some elements like chromium, copper, rhodium, silver and ruthenium do not follow the Aufbau principle. The order of the energy of orbitals can be found out with the help of the $\left(. The arc on orbital chemistry ends with an explanation of two very important rules: the Aufbau Principle, which states that electrons must fill lower energy orbitals before filling higher energy ones, and Hund’s Rules, which state that (1) all subshells must be filled with one electron before being completed by an electron with an opposite spin and (2) the electrons that enter the subshells. In other words, the orbitals having higher energies are filled only after the orbitals with lower energies are completely filled.
It says that the electrons occupy those orbitals first whose energy is the lowest. The order in which atomic orbitals by electrons are filled is based on the Aufbau principle which states that orbitals are filled in increasing energy level. We can understand the Aufbau principle in few points as: The Hund rule: Electrons will occupy dierent orbitals in a given subshell, before two electrons will occupy a single. According to the principle, in the ground state of any atom or ion, the electrons fill the atomic orbitals in increasing order of their energies, that is, filling the lower available energy levels first before the higher levels. The aufbau (building-up) principle: When electrons are lled in to orbitals in an atom, the orbitals with lower energy are lled rst. The Aufbau principle depicts the order in which electrons are filled in the orbitals of an atom in its ground state. Hint: To answer this question, you must recall the Aufbau principle. This gives the following order for filling the orbitals: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p, (8s, 5 g, 6f, 7d, 8p, and.
